Proton gradient drives atp synthesis11/8/2023  
In mitochondria, electron transport drives proton pumping from thematrix into the intermembrane space. Free-energy change during solute movement across a voltage gradient So in chloroplasts, the proton gradient is simply aconcentration gradient, and there is no accompanying voltagegradient.Ĭ. In particular, as H + moves fromthe stroma into the lumen, Mg 2+ moves out of the lumeninto the stroma. The movementof other ions across the thylakoid membrane maintains electricalneutrality across the membrane, despite light-driven proton pumpinginto the thylakoid lumen. This would be the free energyavailable from a gradient of a nonionic solute, or from an ionicgradient if the movement of other ions maintained equal voltage onboth sides of the membranes, as is true in chloroplasts. This is the free-energy change attributable to the concentration gradient. To express DG in terms of the pHgradient (rather than the concentration gradient), change ln to log(that is, log 10) and expand the log term:ĭG = 2.303 RT(log -log) = -2.303 RT(pH in - pH out)ĭ G = - 2.303 RT D pH => (NOTE: DpH = pH in -pH out ) How much free energy is available from the movement of protons downthe concentration gradient created by electron transport?ĭG = DG 0' + RT lnQ = DG 0'+ RTln(/)ĭG 0' = 0 becauseK eq for the process is 1.0 ( DG 0' = - RT ln K eq), Free-energy change during solute movement across a concentration gradientĬonsider the movement of protons from the cytoplasm into the matrixof the mitochondrion: Click here tolearn how to compute activies for solutes, gases, hydrogen ions, andwater or other solvents.ī. Theactivity of the solute is its actual concentration divided by itsstandard concentration, so activities are unitless (which is good,because it's very hard to attach a physical meaning to units like ln ). NOTE: In calculations using this equation, and whenever you haveconcentration terms inside a logarithm term, remember that the properterms are really activities and not concentrations. The actual, or cellular, DGdepends on the cellular ratios of oxidants and reductants:ĭG = DG 0' + RT ln( 2 2)/( 2) cell This process is spontaneous under standard conditions (K eq> 1). Obtain balanced equation for the process described: reverse a) andadd 2 x b):Ģ Cytochrome c-Fe 3+ + 2 e -> 2 Cytochrome c-Fe 2+ 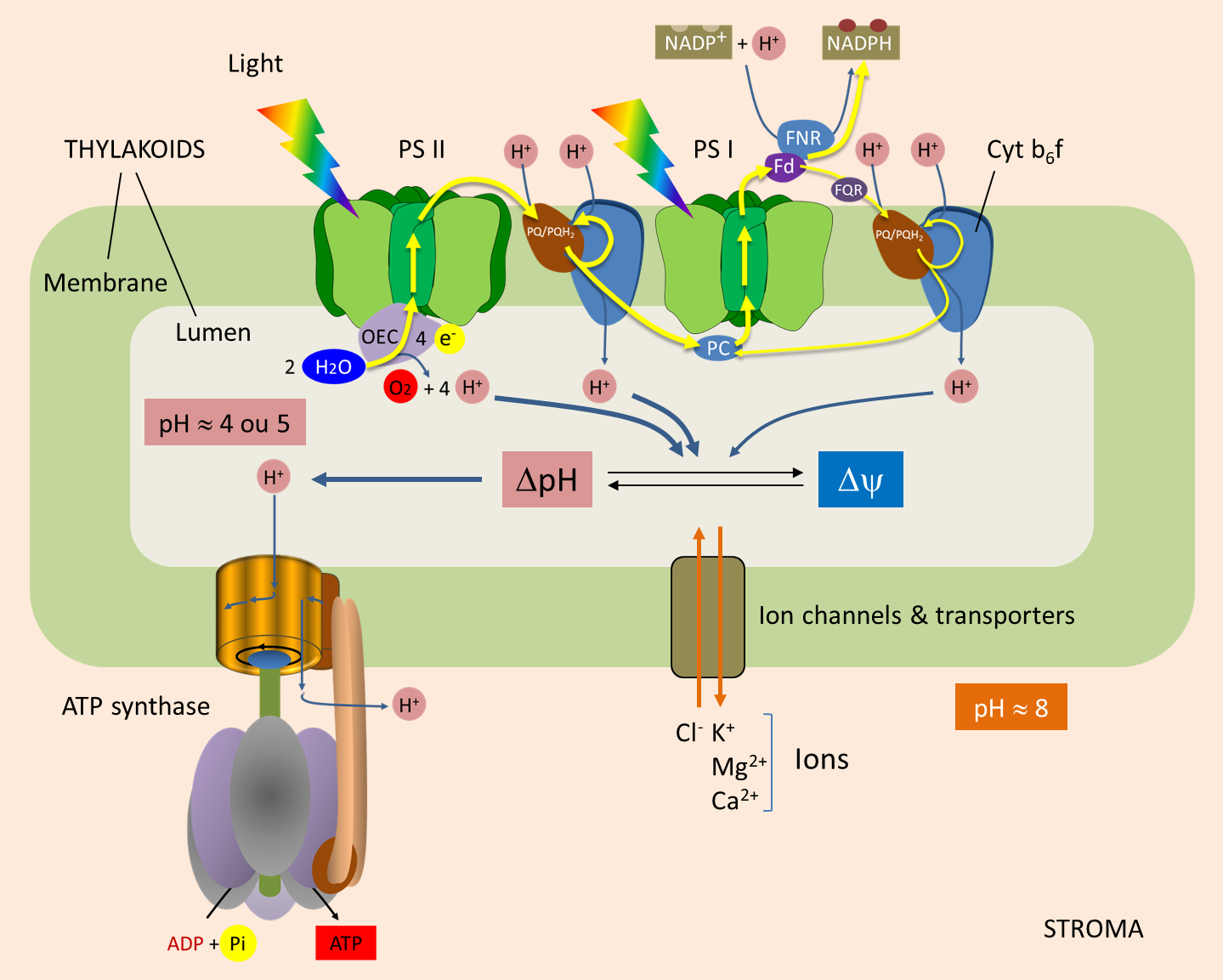
How much freeenergy is available from this process?ī) Cytochrome c-Fe 3+ (ox) + e -> Cytochrome c-Fe 2+ (red) Free-energy change during a redox reactionĬonsider the oxidation of ubiquinone by cytochrome c. I have taken pains in all sections toemploy sign conventions consistently, so take note of how eachprocess is defined, and how the definition determines the sign of thefree-energy change.Ī. This handout shows how to calculate the free energy available fromA) redox reactions B) concentration gradients C) voltage gradients and D) proton or other ion gradients, which have two components,concentration and voltage. (Under construction - watch particularly for omitted minus signs anddeltas, banes of html conversion programs.) Summary: Energy Calculations Mitochondria and Chloroplast Bioenergetics (Terminology Consistent With Lehninger, Nelson, and Cox, Biochemistry, 1993) 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
